Caution: The following texts are translated from the Japanese original. We cannot take responsibility for ambiguity in the translation.
ストナリニS STONARHINI S
Product features
● Stonarhini S has a dual time release structure with the outer layer melting in the stomach and the inner core melting in the intestines, and shows excellent results when taken once or twice daily.
● Datura extract suppresses nasal discharge and shows efficacy toward rhinitis symptoms such as runny nose.
● An easy-to-swallow white sugar-coated tablet.
Indication
Relief of the following symptoms due to acute or allergic rhinitis; sneezing, runny nose, stuffy nose, watery eyes, heaviness in the head
Dosage and administration
[Age: single dose: number of daily doses]
Adult (15 years old and over): 1tablets: 1~2 times
Under 15 years: Do not take.
Precautions of Dosage and Administration
(1)Strictly follow the prescribed dosage and administration.
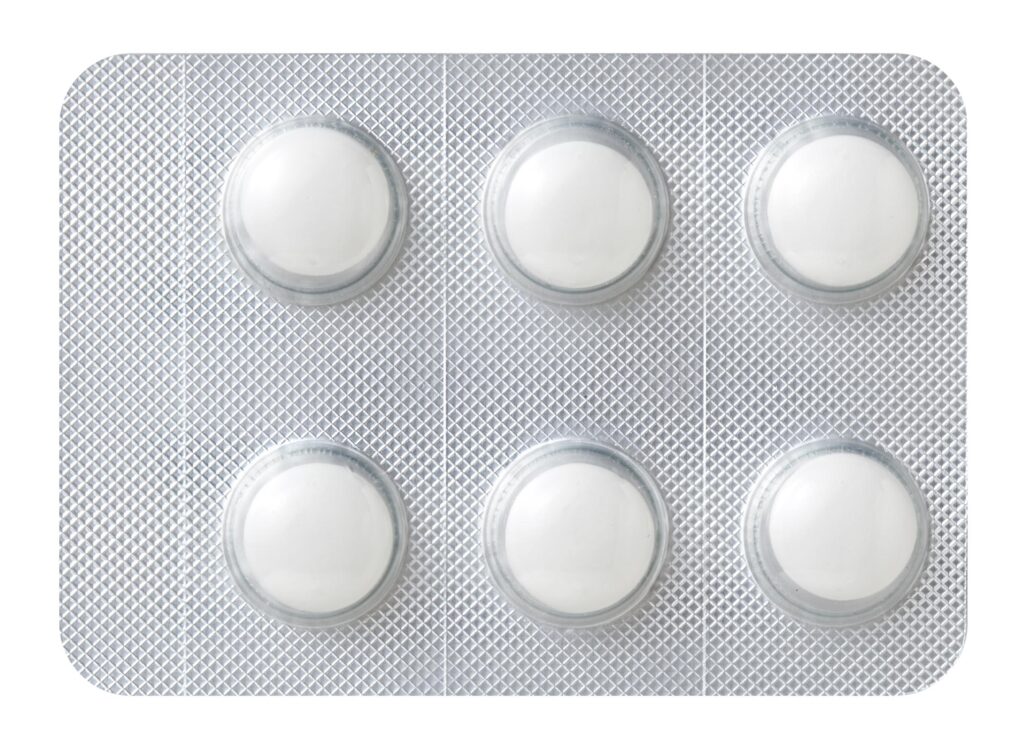
(2)How to Dispense Tablets
Using your finger, push the convex portion of the PTP sheet containing tablets to break the aluminum foil on the back. Please remove the tablet and ingest it.
(Can lead to unforeseen consequences such as piercing the esophageal mucosa if mistakenly swallowed.)
Ingredient and amount
In 2 tablets
<Inner core>
Chlorpheniramine maleate : 6mg: Alleviates allergy related nasal congestion and runny nose.
Phenylephrine Hydrochloride:6mg:Reduces hyperemia (congestion) and swelling in the nasal mucosa and alleviates nasal congestion.
Datura extract:12mg:Works on the parasympathetic nervous system to improve symptoms of rhinitis..
<Outer layer>
Chlorpheniramine maleate : 6mg: Alleviates allergy related nasal congestion and runny nose.
Phenylephrine Hydrochloride:6mg:Reduces hyperemia (congestion) and swelling in the nasal mucosa and alleviates nasal congestion.
Datura extract:12mg: "Works on the parasympathetic nervous system to improve symptoms of rhinitis..
Precautions
■When not to use the product
(If you do not follow these instructions, the current symptoms may worsen or adverse reactions/incidents are more likely to occur.)
1.Do not use any of the following medicines while taking this medicine.
Other internal medicine for rhinitis, internal medicine containing antihistamines (including cold medicine, expectorant, medicine for motion sickness, medicine for allergies), indigestion and heartburn relief.
2.After taking this drug, do not drive a car or operate machinery (symptoms such as sleepiness, blurred vision and abnormal brightness may occur).
3.Do not take this medicine for a long time
■Consultation
1.The following persons should contact a physician, pharmacist, or registered salesperson for a consultation before administration.
(1)Patients undergoing medical treatment from a physician
(2)Pregnant women or women suspected of being pregnant.
(3) The elderly
(4) Patients who have experienced allergic symptoms associated with drugs, etc.
(5) Persons with the following symptoms:
hyperthermia, urination difficulty
(6) Persons diagnosed as having the following
Glaucoma, diabetes, thyroid disease, heart disease, hypertension
2.If the following symptoms are observed after taking this drug, these may be adverse reactions, so immediately discontinue the use of this drug, and show this document to your physician, pharmacist, or registered salesperson for a consultation.
Skin: rash/redness, itching
Gastrointestinal system: nausea and vomiting, loss of appetite
Neuropsychiatric system:headache
Urinary system: urination difficulty
Other:facial hot flashes, abnormal brightness
The following serious symptoms may occur in rare cases. In such cases, immediately seek medical aid:
<Symptom name: Symptoms>
Aplastic anemia: Symptoms, such as bruises, nose bleeds, bleeding gums, fever, pale appearance of skin and mucosa, fatigue, palpitations, shortness of breath, becoming dizzy from feeling sick and blood in the urine, etc. may occur.
Agranulocytosis: Symptoms, such as acute high fever, chills and sore throat, etc. may occur.
3.The following symptoms may be observed after taking this drug. If these symptoms persist or worsen , discontinue the use of this drug, and show this document to your physician, pharmacist, or registered salesperson for a consultation.
dry mouth,drowsiness,constipation,blurred vision
4.When symptoms do not improve even after taking this medicine for 5 to 6 days, stop taking this medicine and consult a physician, pharmacist or registered salesperson, being sure to take this instruction leaflet with you.
Additive
Hydrogen phosphate Ca, lactose, hydroxypropyl cellulose, hydroxypropyl starch, hypromellose, magnesium stearate, hypromellose phthalate, glycerin fatty acid ester, shellac, sucrose, glycerin, talc, polyoxyethylene polyoxypropylene glycol, polyvinyl alcohol (Partially saponified), Ca carbonate, titanium oxide, dimethylpolysiloxane, silicon dioxide, povidone, carnauba wax
Precautions for storage and handling
(1) Store in a cool, low humidity place away from direct sunlight.
(2)Keep out of reach of children.
(3)Do not transfer the medicine to a different container. (It may lead to misuse or alter the quality of the drug.)
(4)Do not take the product after its expiration date.
Disclaimer on Multilingual Product Information
・This product is a pharmaceutical product approved under a Japanese law, the Law for Ensuring the Quality, Efficacy and Safety of Drugs and Medical Devices, with a view to its sale and use in Japan.
・Multilingual product information is a translation of the product labeling written in Japanese and provided for your information only. It does not warrant that its contents and the product itself conforms to laws and regulations in countries other than Japan.
・Multilingual product information is a tentative translation by Our Company, and may be modified or altered without notice.
・Our Company assumes no responsibility for any occurred problem attributable to the contents of the multilingual product information.